Material transfer equipment plays an important role in minimizing contamination risks during the manufacturing process.
Avoiding cross-contamination between rooms with incompatible cleanroom classifications is essential to preserve the desired ISO 14644 classification and maintain the integrity of products and processes.
Non-viable particles and microorganisms can cause contamination in a cleanroom due to this entry and exit of materials within the controlled environments. So with this in mind, it is necessary to design and correctly install material transfer equipment.
A pass box which is also called a transfer hatch or sterile access system, works as a barrier between areas with different levels of cleanliness when materials do have to be moved.
The passbox transfers material from an area of lower cleanliness to an area of higher cleanliness, and vice versa.
All cleanroom passbox include a mechanical and/or electrical interlock system designed to minimize the risk of cross-contamination.
This design ensures that only one door can be opened at a time, not both or more, thus minimizing the amount of “dirty” air that can enter the cleanroom.
There are four main types of pas boxes used in pharma Clean Room.
Dynamic Passbox, (known as the active passbox).
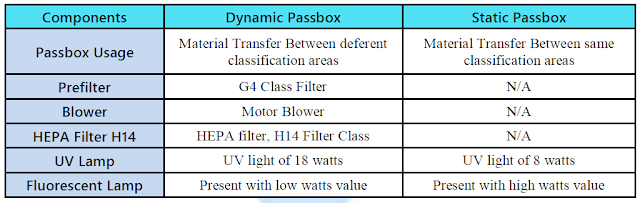
Dynamic passboxs are used to transfer materials from uncontrolled to controlled environments while Static passboxs are used to transfer materials between two rooms of equal cleanliness levels and are designed for minimal personnel movement.
The Dynamic Passbox is installed in the wall between two different grades of clean rooms and is used for materials transfer between areas of low cleanliness to higher cleaned areas or from higher cleaned areas to low cleaned areas.
Mainly the Dynamic Passbox is made of Stainless Steel (SS 304, 316, or 316L)
2. Static Passbox, (known as the passive passbox).
Designed for the transfer of materials between areas with the same cleanliness class, without a filtration system.
The system still uses interlocked doors during the changeover, allowing for the removal of material or transfer of personal from the other door so as not to contaminate the clean area.
The product in transfer is also still subjected to a controlled time of UV light radiation.
3. Ventilated passbox
Designed for the transfer of materials between classified areas or between a classified area and a non-classified area, with decontamination through air sweeping HEPA filters.
The passbox is equipped with a ventilation system coupled with air filtration to ensure a clean environment inside the chamber before the opening of the door.
4. Biological passbox
For the transfer of materials for biological neutralization of elements and bio-contaminated materials between classified areas, or a classified and a non-classified area.
The passbox is equipped with a bio-decontamination system through filtered air and disinfection using a hydrogen peroxide generator (external or integrated)
Usually, passboxs have a UV light lamp in them to remove the contamination that may enter during the transfer of material or personnel.
Note that the passbox can be maintained as a Sink, bubble, or cascade.
As we open the door, an air showering system is activated to remove contamination and to create the required pressure inside the passbox.
Using the dynamic pass is very simple, first of all, open one door and place material inside.
UV light is also turned on when we close the door & the material shall stay as per the validated period, for a validated time period to be properly exposed to UV light
Finally, open the door from the next area to receive the material and after receiving it, close the door.
If a Dynamic Passbox is fitted with a Vaporized Hydrogen Peroxide Generator (VHP) as a decontamination system it may also be called a Bio De-Contamination Passbox.
There is no air showering or air filtration systems in Static pass boxes.
Qualification testing for passbox
Performance qualification shall be performed as per below frequency:
- Filter leakage test (PAO test) – Six Monthly
- Air velocity – Six Monthly
- HEPA pressure difference – Six Monthly
- Particle count – Six Monthly
- Recovery Test – 2 yearly
- Air Visualization Test – 2 yearly
- Noise Test – Six Monthly
- Illumination test – Six Monthly
4.2 Pre-requisites for performance qualification are:
- Inform the concerned department in advance.
- Respective equipment is properly cleaned.
- The pre-filter of RLAF unit are cleaned and Magnehelic gauges are calibrated.
- The instruments for performance qualification are in calibration state and Calibration certificates with traceability to national or international standards are available.
4.3 Filter Integrity test:
4.3.1 Filter leakage test is performed to confirm that the filter system is properly installed and that leaks have not developed during use.
4.3.2 The test shall be performed by introducing the aerosol challenge upstream of the filters and scanning immediately downstream of the filters and support frame.
4.3.3 The test shall be done at rest occupancy states.
4.3.4 Specifications of aerosol photometer: Poly Alpha Olefin (PAO). The concentration of the aerosol shall be 20 – 80 mg/m3. The mass median particle diameter of the aerosol particles shall be between 0.5 to 0.7 µm. The instrument used shall be linear aerosol photometer.
4.3.5 Average reading value for the aerosol shall be provided.
4.3.6 The aerosol concentration measurement shall be taken immediately upstream of the filters and it should not be more than ±15 % than the average measured value.
4.3.7 Probe shall be adequate size.
4.3.8 Probe shall be held in a distance of approximately 3 cm from the downstream filter face or the frame structure.
4.3.9 Scanning shall be performed over the entire downstream face of each filter, the perimeter of each filter, the seal between the filter frame and the grid structure.
4.3.10 Measurement of the aerosol upstream of the filters shall be repeated at reasonable time intervals between and after scanning leaks, to confirm the stability of the challenge aerosol concentration
4.3.11 Acceptance criteria: Leaks are considered to have occurred if the reading is greater than 0.01 % of the upstream challenge aerosol concentration.
4.4 Air Velocity Test:
4.4.1 The purpose of this test is to measure airflow velocity and uniformity and supply air flow rate in the GMP areas.
4.4.2 The supply air flow rate shall be measured downstream of the final filters.
4.4.3 The uniformity of velocity shall be measured at approximately 150 mm to 300mm from the filter face.
4.4.4 The air flow velocity shall be measured with the help of vane type anemometer.
4.4.5 The measuring time at each position should be sufficient to ensure a repeatable reading.
4.4.6 Calculation of air flow supply
Air velocity at each filter = S Air velocity at different locations of filter / Number of locations per filter
Measure the Air velocity at 5 different locations preferably at 4 corners and at the centre of the filter.
4.4.7 Acceptance Criteria : 90 ± 20 feet per minute.
4.5 HEPA filter pressure difference test:
4.5.1 The purpose of this test is to verify the capability of complete installation to maintain specific pressure differentials across HEPA.
4.5.2 Check and record the pressure difference across HEPA
4.5.3 Acceptance criteria: – Between 8 mm – 15 mm of WG
4.6 Air borne particulate count:
4.6.1 Air borne particle counting is done to measure the airborne particle count concentrations of size 0.5 mm and 5 mm.
4.6.2 Check that following tests are passing before starting airborne particle count:
- Airflow velocity
- Installed filter leakage test
- HEPA filter pressure difference test
4.6.3 Particle count shall be checked at 3 locations per filter for RLAF and LAF and 1 location per filter for Dynamic Pass Box at the height of the work activity. The particle counter shall be set to provided print with
- Instrument details
- Date and time
- Measurement of particle count ranging from 0.3 µm to 25.0 µm.
- 3 consecutive readings for 1 minute shall be taken and
- Average at each location
4.6.4 Acceptance criteria: (At rest)
| ISO classification number | Maximum concentration limit (Particles / m3) | |
| 0.5 mm | 5.0 mm | |
| ISO class 5 | 3,520 | 29 |
4.7 Recovery Test:
4.7.1 Recovery test is done to find out the recovery time of the LAF, RLAF after stoppage or break down.
4.7.2 Particle size used in this test shall be same as that used for the determination of cleanliness class.
4.7.3 Sampling Location of the room showing maximum count shall be selected for monitoring the recovery time.
4.7.4 A duplicate reading shall be taken at this location while the equipment is in ON condition. The counter is maintained in running mode. The equipment is then put off. The Particle count for each minute is taken until the clean room condition is disturbed .i.e. the particle count exceeds clean room specification limits. The time period required to disturbed the cleanliness condition is noted. (This is HOLD Time)
4.7.5 The Equipment is restarted; the particle count is taken every minute from the start.
4.7.6 The time noted at which the particulate count regains the clean room specification.
4.8 Air Visualization pattern (air flow pattern test):
4.8.1 This test shall be done to demonstrate the Laminar pattern of the air supplied. A recorded flow pattern shall be maintained as CD.
4.9 Noise and Illumination Test: This test shall be done to demonstrate the Noise level and light illumination of the equipment.
4.9.1 Noise level shall be checked by using the sound level meter. Check the calibration status of the instrument before use. The noise level should not be more than 85 dB.
4.9.2 Light illumination shall be checked by using a lux meter. Check the calibration status of the instrument before use. The Lux level should not be less than 150 lux.
4.10 After completion of the activity, raw data shall be evaluated by engineering and QA personnel. If the results are found to be within the specified limits, affix qualification status labels.














.JPG)





0 Comments